 Gene counts are sourced from ARCHS4, which provides uniform alignment of GEO samples.
You can learn more about ARCHS4 and its pipeline here.
Gene counts are sourced from ARCHS4, which provides uniform alignment of GEO samples.
You can learn more about ARCHS4 and its pipeline here.
Select conditions below to toggle them from the plot:
| GROUP | CONDITION | SAMPLES |
|---|---|---|
| KR mutant |
GSM2055667
|
|
|
GSM2055668
|
||
| WT |
GSM2055666
|
|
|
GSM2055669
|
Submission Date: Feb 08, 2016
Summary: Pancreatic beta-cell dysfunction contributes to onset and progression of type 2 diabetes. In this state beta-cells become metabolically inflexible, losing the ability to select between carbohydrates and lipids as substrates for mitochondrial oxidation. These changes lead to beta-cell dedifferentiation. We have proposed that FoxO proteins are activated through deacetylation-dependent nuclear translocation to forestall the progression of these abnormalities. However, how deacetylated FoxO exert their actions remains unclear. To address this question, we analyzed islet function in mice homozygous for knock-in alleles encoding deacetylated FoxO1 (6KR). Islets expressing 6KR mutant FoxO1 have enhanced insulin secretion in vivo and ex vivo, and decreased fatty acid oxidation ex vivo. Remarkably, the gene expression signature associated with FoxO1 deacetylation differs from wild-type by only ~2% of the > 4,000 genes regulated in response to re-feeding. But this narrow swath includes key genes required for beta-cell identity, lipid metabolism, and mitochondrial fatty acid and solute transport. The data support the notion that deacetylated FoxO1 protects beta-cell function by limiting mitochondrial lipid utilization, and raise the possibility that inhibition of fatty acid oxidation in β-cells is beneficial to diabetes treatment.
GEO Accession ID: GSE77652
PMID: 26984405
Submission Date: Feb 08, 2016
Summary: Pancreatic beta-cell dysfunction contributes to onset and progression of type 2 diabetes. In this state beta-cells become metabolically inflexible, losing the ability to select between carbohydrates and lipids as substrates for mitochondrial oxidation. These changes lead to beta-cell dedifferentiation. We have proposed that FoxO proteins are activated through deacetylation-dependent nuclear translocation to forestall the progression of these abnormalities. However, how deacetylated FoxO exert their actions remains unclear. To address this question, we analyzed islet function in mice homozygous for knock-in alleles encoding deacetylated FoxO1 (6KR). Islets expressing 6KR mutant FoxO1 have enhanced insulin secretion in vivo and ex vivo, and decreased fatty acid oxidation ex vivo. Remarkably, the gene expression signature associated with FoxO1 deacetylation differs from wild-type by only ~2% of the > 4,000 genes regulated in response to re-feeding. But this narrow swath includes key genes required for beta-cell identity, lipid metabolism, and mitochondrial fatty acid and solute transport. The data support the notion that deacetylated FoxO1 protects beta-cell function by limiting mitochondrial lipid utilization, and raise the possibility that inhibition of fatty acid oxidation in β-cells is beneficial to diabetes treatment.
GEO Accession ID: GSE77652
PMID: 26984405
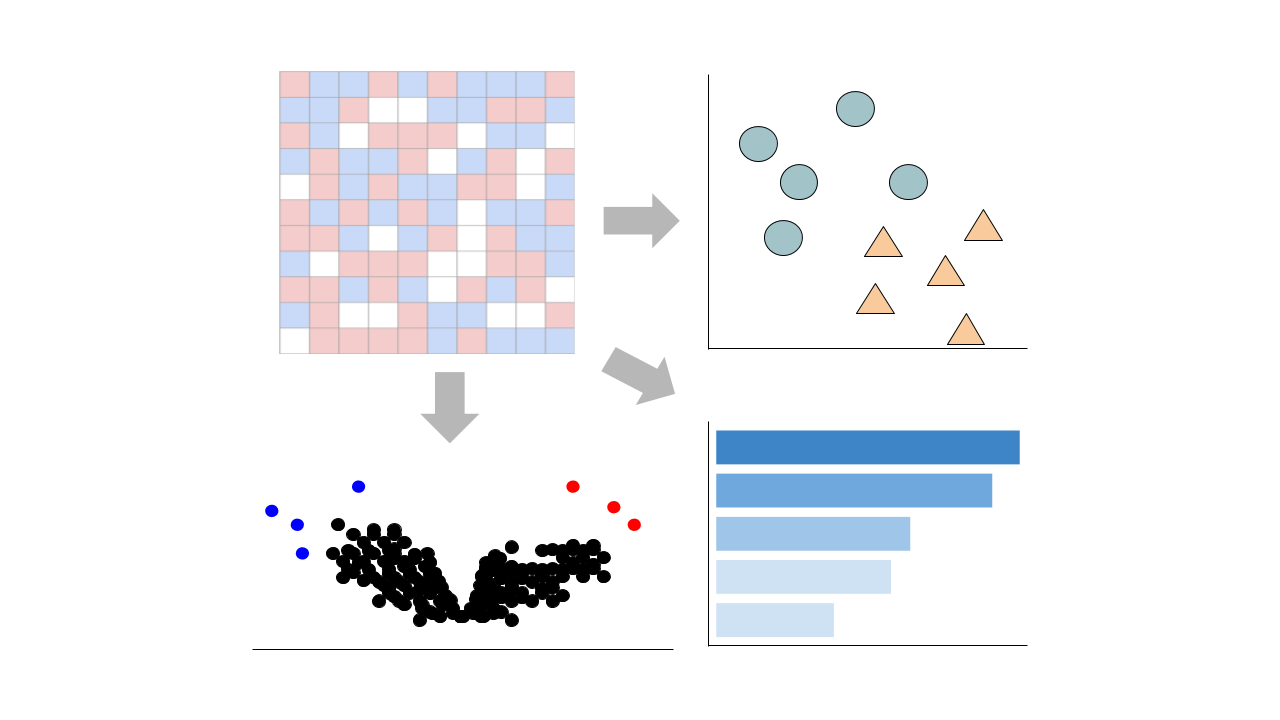
Visualize Samples
 Visualizations are precomputed using the Python package scanpy on the top 5000 most variable genes.
Visualizations are precomputed using the Python package scanpy on the top 5000 most variable genes.
Precomputed Differential Gene Expression
 Differential expression signatures are automatically computed using the limma R package.
More options for differential expression are available to compute below.
Differential expression signatures are automatically computed using the limma R package.
More options for differential expression are available to compute below.
Signatures:
No precomputed signatures are currently available for this study. You can compute differential gene expression on the fly below:
Select conditions:
Control Condition
Perturbation Condition
Only conditions with at least 1 replicate are available to select
 Differential expression signatures can be computed using DESeq2 or characteristic direction.
Differential expression signatures can be computed using DESeq2 or characteristic direction.
This pipeline enables you to analyze and visualize your bulk RNA sequencing datasets with an array of downstream analysis and visualization tools. The pipeline includes: PCA analysis, Clustergrammer interactive heatmap, library size analysis, differential gene expression analysis, enrichment analysis, and L1000 small molecule search.
 Chatbot
Chatbot Single Gene Queries
Single Gene Queries
 Gene Set Queries
Gene Set Queries
 Bulk Studies
Bulk Studies
 Single Cell Studies
Single Cell Studies
 Hypotheses
Hypotheses
 Resources
Resources
 Contribute
Contribute
 Downloads
Downloads About
About
 Help
Help