 Gene counts are sourced from ARCHS4, which provides uniform alignment of GEO samples.
You can learn more about ARCHS4 and its pipeline here.
Gene counts are sourced from ARCHS4, which provides uniform alignment of GEO samples.
You can learn more about ARCHS4 and its pipeline here.
Select conditions below to toggle them from the plot:
| GROUP | CONDITION | SAMPLES |
|---|---|---|
| blood |
GSM4799154 GSM4799156 GSM4799158 GSM4799160 GSM4799162 GSM4799164 GSM4799166 GSM4799168 GSM4799170 GSM4799172 GSM4799174 GSM4799176 GSM4799178 GSM4799180 GSM4799182 GSM4799184 GSM4799186 GSM4799188 GSM4799190 GSM4799192 GSM4799194 GSM4799196 GSM4799198 GSM4799200 GSM4799202 GSM4799204 GSM4799206 GSM4799208
|
|
|
GSM4799153 GSM4799155 GSM4799157 GSM4799159 GSM4799161 GSM4799163 GSM4799165 GSM4799167 GSM4799169 GSM4799171 GSM4799173 GSM4799175 GSM4799177 GSM4799179 GSM4799181 GSM4799183 GSM4799185 GSM4799187 GSM4799189 GSM4799191 GSM4799193 GSM4799195 GSM4799197 GSM4799199 GSM4799201 GSM4799203 GSM4799205 GSM4799207
|
Submission Date: Sep 23, 2020
Summary: Purpose: We aim to identify transciptional changes of human CD4+/CD8+ T cells due to high fat low carbohydrate ketogenic diet (KD) in vivo.
Method: Healthy volunteers conducted a 21 days ketogenic diet, limiting carbohydrate intake to <30g/day. Before the start (T0) and at the end (T1) of the diet, blood samples were taken and PBMC were isolated. PBMCs were obtained by density centrifugation (Histopaque 1077, Sigma-Aldrich, St. Louis, MO, USA). A ViCell analyzer (Beckman Coulter, Fullerton, CA, USA) was used to evaluate the cell count and viability. Only samples exceeding a cell viability of 90% were processed further. PBMCs were subjected to cell cultivation in RPMI 1640 (Invitrogen, Carlsbad, CA, USA) containing 10% heat-inactivated fetal calf serum (Biochrom, Berlin, Germany), 1% HEPES (Sigma-Aldrich, St. Louis, MO) and 1% L-glutamine (Life Technologies, Carlsbad, CA, USA). T cells were stimulated via the addition of CD3/CD28 Dynabeads (Thermo Fisher Scientific, Waltham, MA, USA) with a bead-to-cell ratio of 1:8 for a duration 24 hours. After stimulation, CD3/CD28 Dynabeads were magnetically removed. Pan T cell-, CD4+- and CD8+-cell-isolation was performed by magnetic cell separation (Pan T Cell Isolation Kit, # 130-096-535 | human CD4 MicroBeads, # 130-045-101 | human CD8 MicroBeads, # 130-045-201, Miltenyi Biotec, Bergisch Gladbach, Germany) using an AutoMACS Pro Separator ( # 130-092-545, Miltenyi Biotec, Bergisch-Gladbach, Germany) according to the manufacturer's instructions.
Results: 11.545 expressed genes were identified, for CD4/CD8 T cells, we detected 5.667/ 5.799 up-regulated genes and 5.878/5.746 down-regulated genes. 294/346 genes and 325/252 genes were significantly up/down-regulated for CD4+/CD8+ T cells (p-val. <0.05). Gene set enrichment analysis revealed 117/17 and 22/6 significantly up/down-regulated pathways for CD4+/CD8+ T cells (p-val. <0.05). Genes and Gene sets differentially regulated were relevant for T cell immune response and metabolic function.
Conclusion: KD resulted in immunometabolic reprogramming of human CD4+/CD8+ T cells.
GEO Accession ID: GSE158407
PMID: 34151532
Submission Date: Sep 23, 2020
Summary: Purpose: We aim to identify transciptional changes of human CD4+/CD8+ T cells due to high fat low carbohydrate ketogenic diet (KD) in vivo.
Method: Healthy volunteers conducted a 21 days ketogenic diet, limiting carbohydrate intake to <30g/day. Before the start (T0) and at the end (T1) of the diet, blood samples were taken and PBMC were isolated. PBMCs were obtained by density centrifugation (Histopaque 1077, Sigma-Aldrich, St. Louis, MO, USA). A ViCell analyzer (Beckman Coulter, Fullerton, CA, USA) was used to evaluate the cell count and viability. Only samples exceeding a cell viability of 90% were processed further. PBMCs were subjected to cell cultivation in RPMI 1640 (Invitrogen, Carlsbad, CA, USA) containing 10% heat-inactivated fetal calf serum (Biochrom, Berlin, Germany), 1% HEPES (Sigma-Aldrich, St. Louis, MO) and 1% L-glutamine (Life Technologies, Carlsbad, CA, USA). T cells were stimulated via the addition of CD3/CD28 Dynabeads (Thermo Fisher Scientific, Waltham, MA, USA) with a bead-to-cell ratio of 1:8 for a duration 24 hours. After stimulation, CD3/CD28 Dynabeads were magnetically removed. Pan T cell-, CD4+- and CD8+-cell-isolation was performed by magnetic cell separation (Pan T Cell Isolation Kit, # 130-096-535 | human CD4 MicroBeads, # 130-045-101 | human CD8 MicroBeads, # 130-045-201, Miltenyi Biotec, Bergisch Gladbach, Germany) using an AutoMACS Pro Separator ( # 130-092-545, Miltenyi Biotec, Bergisch-Gladbach, Germany) according to the manufacturer's instructions.
Results: 11.545 expressed genes were identified, for CD4/CD8 T cells, we detected 5.667/ 5.799 up-regulated genes and 5.878/5.746 down-regulated genes. 294/346 genes and 325/252 genes were significantly up/down-regulated for CD4+/CD8+ T cells (p-val. <0.05). Gene set enrichment analysis revealed 117/17 and 22/6 significantly up/down-regulated pathways for CD4+/CD8+ T cells (p-val. <0.05). Genes and Gene sets differentially regulated were relevant for T cell immune response and metabolic function.
Conclusion: KD resulted in immunometabolic reprogramming of human CD4+/CD8+ T cells.
GEO Accession ID: GSE158407
PMID: 34151532
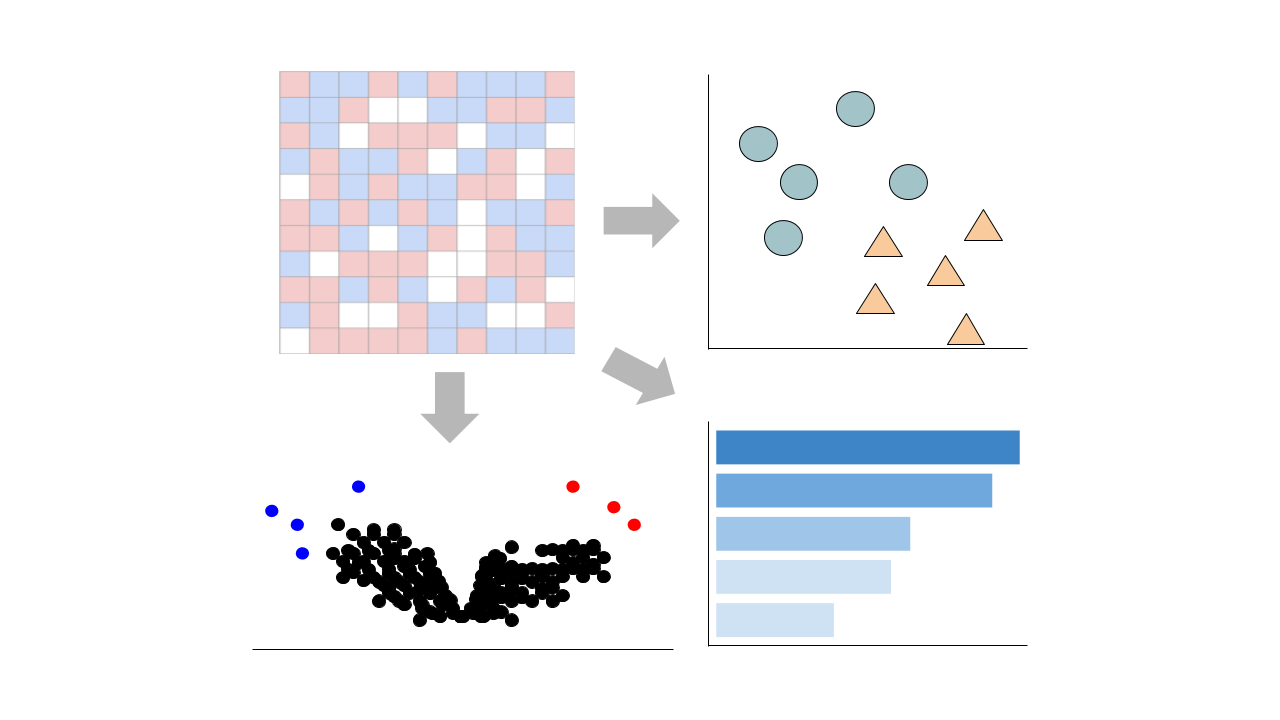
Visualize Samples
 Visualizations are precomputed using the Python package scanpy on the top 5000 most variable genes.
Visualizations are precomputed using the Python package scanpy on the top 5000 most variable genes.
Precomputed Differential Gene Expression
 Differential expression signatures are automatically computed using the limma R package.
More options for differential expression are available to compute below.
Differential expression signatures are automatically computed using the limma R package.
More options for differential expression are available to compute below.
Signatures:
Select conditions:
Control Condition
Perturbation Condition
Only conditions with at least 1 replicate are available to select
 Differential expression signatures can be computed using DESeq2 or characteristic direction.
Differential expression signatures can be computed using DESeq2 or characteristic direction.
This pipeline enables you to analyze and visualize your bulk RNA sequencing datasets with an array of downstream analysis and visualization tools. The pipeline includes: PCA analysis, Clustergrammer interactive heatmap, library size analysis, differential gene expression analysis, enrichment analysis, and L1000 small molecule search.
 Chatbot
Chatbot Single Gene Queries
Single Gene Queries
 Gene Set Queries
Gene Set Queries
 Bulk Studies
Bulk Studies
 Single Cell Studies
Single Cell Studies
 Hypotheses
Hypotheses
 Resources
Resources
 Contribute
Contribute
 Downloads
Downloads About
About
 Help
Help