 Gene counts are sourced from ARCHS4, which provides uniform alignment of GEO samples.
You can learn more about ARCHS4 and its pipeline here.
Gene counts are sourced from ARCHS4, which provides uniform alignment of GEO samples.
You can learn more about ARCHS4 and its pipeline here.
Select conditions below to toggle them from the plot:
| GROUP | CONDITION | SAMPLES |
|---|---|---|
| High-fat diet for 8 months; 129/B6 mice, age 10 months |
GSM4718414 GSM4718415 GSM4718416
|
|
|
GSM4718411 GSM4718412 GSM4718413
|
||
| Low-fat diet for 8 months; 129/B6 mice, age 10 months |
GSM4718408 GSM4718409 GSM4718410
|
|
|
GSM4718405 GSM4718406 GSM4718407
|
Submission Date: Aug 10, 2020
Summary: Thrombospondin 1 (TSP1) is a multifunctional matricellular protein. Previously we have shown that TSP1 plays an important role in obesity-associated metabolic complications including inflammation, insulin resistance, cardiovascular and renal disease. However, its contribution to obesity-associated non-alcoholic fatty liver disease (NAFLD) or non-alcoholic steatohepatitis (NASH) remains largely unknown and is determined in this study. High fat diet or AMLN diet-induced obese and insulin resistant NAFLD/NASH mouse models were utilized. In addition, tissue specific TSP1 knockout mice were utilized to determine the contribution of different cellular sources of obesity-induced TSP1 to NAFLD/NASH development. The data demonstrated that liver TSP1 levels were increased in experimental obese and insulin resistant NAFLD/NASH mouse models as well as in human obese NASH patients. Moreover, TSP1 deletion in hepatocyte or adipocytes did not protect mice from diet-induced NAFLD/NASH. However, myeloid/macrophage-specific TSP1 deletion protected mice against obesity-associated liver injury, accompanied by reduced liver inflammation and fibrosis. Importantly, this protection is independent of the levels of obesity and hepatic steatosis. Mechanistically, through an autocrine effect, macrophage-derived TSP1 suppressed SMPDL3B expression in liver, which amplified liver pro-inflammatory signaling (TLR4 signal pathway) and promoted NAFLD progression. Together, out data suggest that macrophage-derived TSP1 is a significant contributor to obesity-associated NAFLD/NASH development and progression and may serve as a therapeutic target for this disease.
GEO Accession ID: GSE155973
PMID: 33294831
Submission Date: Aug 10, 2020
Summary: Thrombospondin 1 (TSP1) is a multifunctional matricellular protein. Previously we have shown that TSP1 plays an important role in obesity-associated metabolic complications including inflammation, insulin resistance, cardiovascular and renal disease. However, its contribution to obesity-associated non-alcoholic fatty liver disease (NAFLD) or non-alcoholic steatohepatitis (NASH) remains largely unknown and is determined in this study. High fat diet or AMLN diet-induced obese and insulin resistant NAFLD/NASH mouse models were utilized. In addition, tissue specific TSP1 knockout mice were utilized to determine the contribution of different cellular sources of obesity-induced TSP1 to NAFLD/NASH development. The data demonstrated that liver TSP1 levels were increased in experimental obese and insulin resistant NAFLD/NASH mouse models as well as in human obese NASH patients. Moreover, TSP1 deletion in hepatocyte or adipocytes did not protect mice from diet-induced NAFLD/NASH. However, myeloid/macrophage-specific TSP1 deletion protected mice against obesity-associated liver injury, accompanied by reduced liver inflammation and fibrosis. Importantly, this protection is independent of the levels of obesity and hepatic steatosis. Mechanistically, through an autocrine effect, macrophage-derived TSP1 suppressed SMPDL3B expression in liver, which amplified liver pro-inflammatory signaling (TLR4 signal pathway) and promoted NAFLD progression. Together, out data suggest that macrophage-derived TSP1 is a significant contributor to obesity-associated NAFLD/NASH development and progression and may serve as a therapeutic target for this disease.
GEO Accession ID: GSE155973
PMID: 33294831
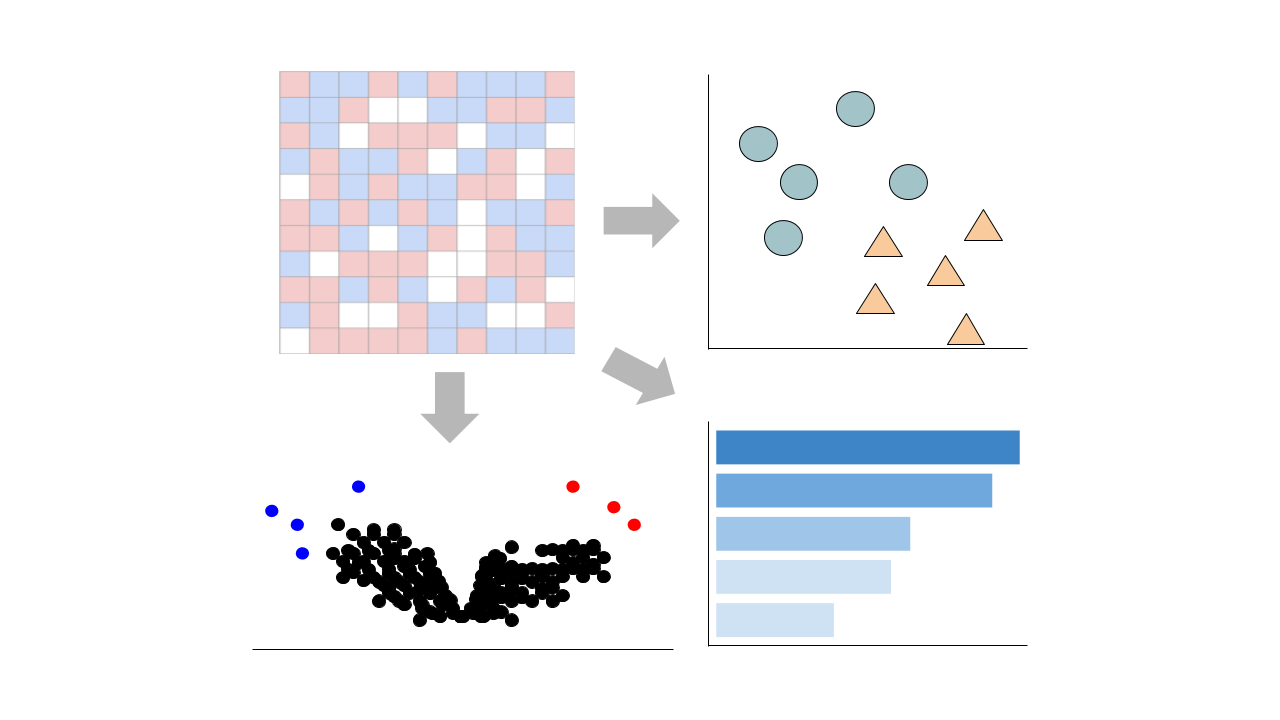
Visualize Samples
 Visualizations are precomputed using the Python package scanpy on the top 5000 most variable genes.
Visualizations are precomputed using the Python package scanpy on the top 5000 most variable genes.
Precomputed Differential Gene Expression
 Differential expression signatures are automatically computed using the limma R package.
More options for differential expression are available to compute below.
Differential expression signatures are automatically computed using the limma R package.
More options for differential expression are available to compute below.
Signatures:
Select conditions:
Control Condition
Perturbation Condition
Only conditions with at least 1 replicate are available to select
 Differential expression signatures can be computed using DESeq2 or characteristic direction.
Differential expression signatures can be computed using DESeq2 or characteristic direction.
This pipeline enables you to analyze and visualize your bulk RNA sequencing datasets with an array of downstream analysis and visualization tools. The pipeline includes: PCA analysis, Clustergrammer interactive heatmap, library size analysis, differential gene expression analysis, enrichment analysis, and L1000 small molecule search.
 Chatbot
Chatbot Single Gene Queries
Single Gene Queries
 Gene Set Queries
Gene Set Queries
 Bulk Studies
Bulk Studies
 Single Cell Studies
Single Cell Studies
 Hypotheses
Hypotheses
 Resources
Resources
 Contribute
Contribute
 Downloads
Downloads About
About
 Help
Help