 Gene counts are sourced from ARCHS4, which provides uniform alignment of GEO samples.
You can learn more about ARCHS4 and its pipeline here.
Gene counts are sourced from ARCHS4, which provides uniform alignment of GEO samples.
You can learn more about ARCHS4 and its pipeline here.
Select conditions below to toggle them from the plot:
| GROUP | CONDITION | SAMPLES |
|---|---|---|
| Pancreatic islet-derived exosome |
GSM4149721 GSM4149723 GSM4149725 GSM4149727 GSM4149729
|
|
|
GSM4149722 GSM4149724 GSM4149726 GSM4149728 GSM4149730
|
Submission Date: Nov 05, 2019
Summary: Type 1 diabetes (T1D) is characterized by immune mediated destruction of insulin producing β cells. Biomarkers capable of identifying T1D risk and dissecting disease-related heterogeneity represent an unmet clinical need. Aims: Towards the goal of informing T1D biomarker strategies, we profiled different classes of RNAs in human islet-derived exosomes and identified RNAs that were differentially expressed under cytokine stress conditions. Human pancreatic islets were obtained from cadaveric donors and treated with/without IL-1β and IFN-γ to mimic the pro-inflammatory T1D milieu. Total RNA and small RNA sequencing were performed to identify long (mRNA and long non-coding RNAs) and different classes of small non-coding RNAs. RNAs with fold change ≥ 1.3 and p-value < 0.05 were considered as differentially expressed. mRNAs and miRNAs species represented the most abundant long and small RNA species, respectively. Expression patterns of each class of RNA were changed with cytokine treatment. Differentially expressed long RNAs and targets of small non-coding RNAs were predicted to be involved in insulin secretion, calcium signaling, necrosis and apoptosis. Our data provides the first comprehensive catalog of protein coding and non-coding RNAs in human islet-derived exosomes and identifies RNAs that are dysregulated under cytokine stress.
GEO Accession ID: GSE139932
PMID: 31775218
Submission Date: Nov 05, 2019
Summary: Type 1 diabetes (T1D) is characterized by immune mediated destruction of insulin producing β cells. Biomarkers capable of identifying T1D risk and dissecting disease-related heterogeneity represent an unmet clinical need. Aims: Towards the goal of informing T1D biomarker strategies, we profiled different classes of RNAs in human islet-derived exosomes and identified RNAs that were differentially expressed under cytokine stress conditions. Human pancreatic islets were obtained from cadaveric donors and treated with/without IL-1β and IFN-γ to mimic the pro-inflammatory T1D milieu. Total RNA and small RNA sequencing were performed to identify long (mRNA and long non-coding RNAs) and different classes of small non-coding RNAs. RNAs with fold change ≥ 1.3 and p-value < 0.05 were considered as differentially expressed. mRNAs and miRNAs species represented the most abundant long and small RNA species, respectively. Expression patterns of each class of RNA were changed with cytokine treatment. Differentially expressed long RNAs and targets of small non-coding RNAs were predicted to be involved in insulin secretion, calcium signaling, necrosis and apoptosis. Our data provides the first comprehensive catalog of protein coding and non-coding RNAs in human islet-derived exosomes and identifies RNAs that are dysregulated under cytokine stress.
GEO Accession ID: GSE139932
PMID: 31775218
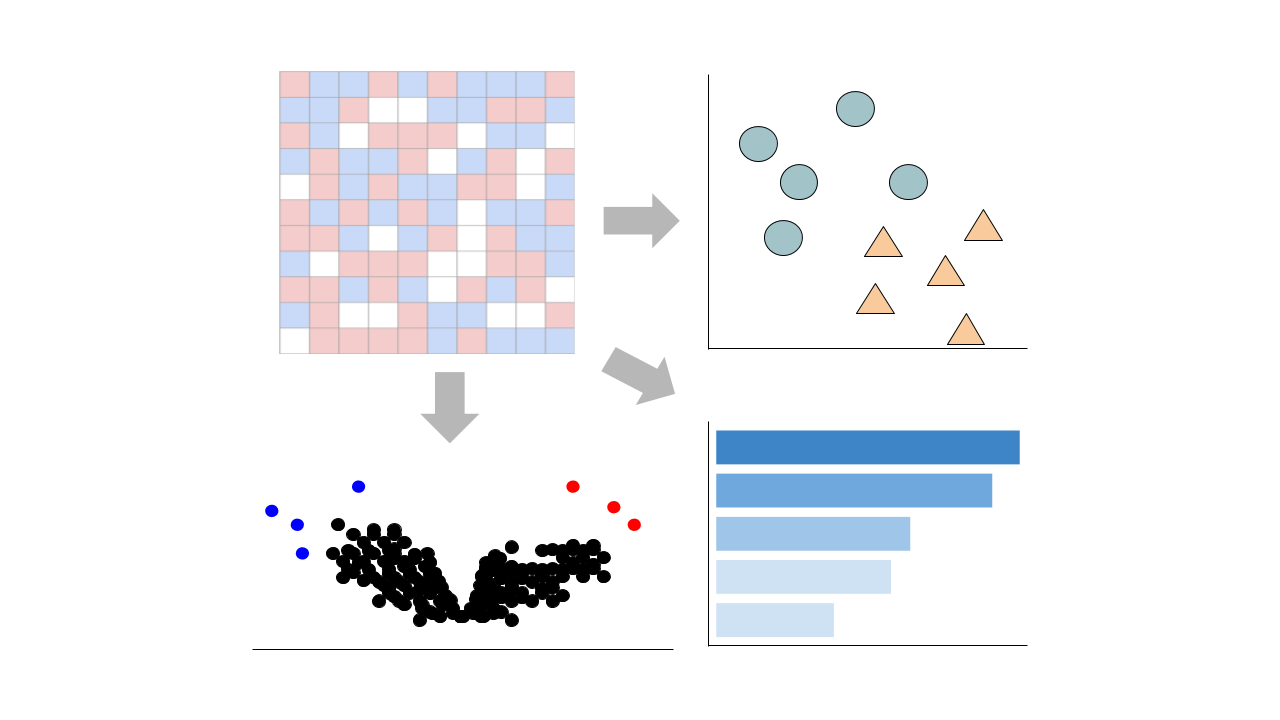
Visualize Samples
 Visualizations are precomputed using the Python package scanpy on the top 5000 most variable genes.
Visualizations are precomputed using the Python package scanpy on the top 5000 most variable genes.
Precomputed Differential Gene Expression
 Differential expression signatures are automatically computed using the limma R package.
More options for differential expression are available to compute below.
Differential expression signatures are automatically computed using the limma R package.
More options for differential expression are available to compute below.
Signatures:
Select conditions:
Control Condition
Perturbation Condition
Only conditions with at least 1 replicate are available to select
 Differential expression signatures can be computed using DESeq2 or characteristic direction.
Differential expression signatures can be computed using DESeq2 or characteristic direction.
This pipeline enables you to analyze and visualize your bulk RNA sequencing datasets with an array of downstream analysis and visualization tools. The pipeline includes: PCA analysis, Clustergrammer interactive heatmap, library size analysis, differential gene expression analysis, enrichment analysis, and L1000 small molecule search.
 Chatbot
Chatbot Single Gene Queries
Single Gene Queries
 Gene Set Queries
Gene Set Queries
 Bulk Studies
Bulk Studies
 Single Cell Studies
Single Cell Studies
 Hypotheses
Hypotheses
 Resources
Resources
 Contribute
Contribute
 Downloads
Downloads About
About
 Help
Help