 Gene counts are sourced from ARCHS4, which provides uniform alignment of GEO samples.
You can learn more about ARCHS4 and its pipeline here.
Gene counts are sourced from ARCHS4, which provides uniform alignment of GEO samples.
You can learn more about ARCHS4 and its pipeline here.
Select conditions below to toggle them from the plot:
| GROUP | CONDITION | SAMPLES |
|---|---|---|
| Perigonadal Whole Adipose Tissue |
GSM3711931 GSM3711932 GSM3711933 GSM3711934
|
|
|
GSM3711935 GSM3711936 GSM3711937 GSM3711938
|
Submission Date: Apr 04, 2019
Summary: The Silencing Mediator of Retinoid and Thyroid Hormone Receptors (SMRT) is a nuclear corepressor that regulates the transcriptional activity of many transcription factors critical for metabolic processes. While the importance of SMRT's role in the adipocyte has been well-established, prior mouse models have yielded contradictory phenotypes, limiting our understanding of its in vivo function in the context of homeostatic maintenance. Multiple models suggest that SMRT deficiency leads to increased adiposity, though the effects of SMRT loss on glucose tolerance and insulin sensitivity have been variable. We therefore generated an adipocyte-specific SMRT knockout (adSMRT-/-) mouse to more clearly define SMRT's metabolic contributions. In doing so, we found that SMRT deletion in the adipocyte does not, in fact, lead to obesity, despite increased food consumption in knockouts – even when mice are challenged with a high-fat diet. This suggests that prior adiposity phenotypes described in generalized models were due to effects beyond the adipocyte. However, an adipocyte-specific SMRT deficiency still led to dramatic effects on systemic glucose tolerance and adipocyte insulin sensitivity, impairing both. This metabolically deleterious effect was coupled with a surprising immune phenotype, wherein most genes differentially expressed in the adipose tissue of adSMRT-/- mice were upregulated in pro-inflammatory pathways. Flow cytometry and conditioned media experiments demonstrated that secreted factors from knockout adipose tissue strongly informed resident macrophages to develop a pro-inflammatory, MMe (metabolically activated) phenotype. Taken together, these studies suggest a novel role for SMRT as an integrator of metabolic and inflammatory signals to effectively maintain physiological homeostasis.
GEO Accession ID: GSE129369
PMID: No Pubmed ID
Submission Date: Apr 04, 2019
Summary: The Silencing Mediator of Retinoid and Thyroid Hormone Receptors (SMRT) is a nuclear corepressor that regulates the transcriptional activity of many transcription factors critical for metabolic processes. While the importance of SMRT's role in the adipocyte has been well-established, prior mouse models have yielded contradictory phenotypes, limiting our understanding of its in vivo function in the context of homeostatic maintenance. Multiple models suggest that SMRT deficiency leads to increased adiposity, though the effects of SMRT loss on glucose tolerance and insulin sensitivity have been variable. We therefore generated an adipocyte-specific SMRT knockout (adSMRT-/-) mouse to more clearly define SMRT's metabolic contributions. In doing so, we found that SMRT deletion in the adipocyte does not, in fact, lead to obesity, despite increased food consumption in knockouts – even when mice are challenged with a high-fat diet. This suggests that prior adiposity phenotypes described in generalized models were due to effects beyond the adipocyte. However, an adipocyte-specific SMRT deficiency still led to dramatic effects on systemic glucose tolerance and adipocyte insulin sensitivity, impairing both. This metabolically deleterious effect was coupled with a surprising immune phenotype, wherein most genes differentially expressed in the adipose tissue of adSMRT-/- mice were upregulated in pro-inflammatory pathways. Flow cytometry and conditioned media experiments demonstrated that secreted factors from knockout adipose tissue strongly informed resident macrophages to develop a pro-inflammatory, MMe (metabolically activated) phenotype. Taken together, these studies suggest a novel role for SMRT as an integrator of metabolic and inflammatory signals to effectively maintain physiological homeostasis.
GEO Accession ID: GSE129369
PMID: No Pubmed ID
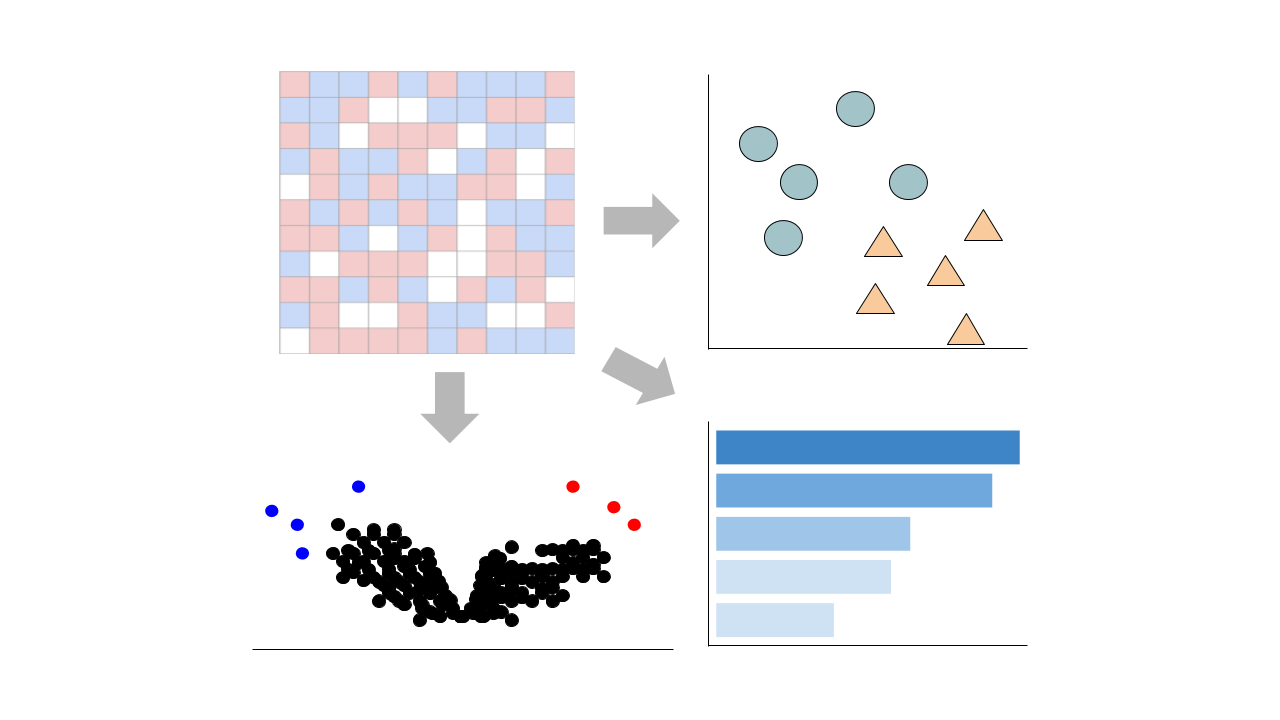
Visualize Samples
 Visualizations are precomputed using the Python package scanpy on the top 5000 most variable genes.
Visualizations are precomputed using the Python package scanpy on the top 5000 most variable genes.
Precomputed Differential Gene Expression
 Differential expression signatures are automatically computed using the limma R package.
More options for differential expression are available to compute below.
Differential expression signatures are automatically computed using the limma R package.
More options for differential expression are available to compute below.
Signatures:
Select conditions:
Control Condition
Perturbation Condition
Only conditions with at least 1 replicate are available to select
 Differential expression signatures can be computed using DESeq2 or characteristic direction.
Differential expression signatures can be computed using DESeq2 or characteristic direction.
This pipeline enables you to analyze and visualize your bulk RNA sequencing datasets with an array of downstream analysis and visualization tools. The pipeline includes: PCA analysis, Clustergrammer interactive heatmap, library size analysis, differential gene expression analysis, enrichment analysis, and L1000 small molecule search.
 Chatbot
Chatbot Single Gene Queries
Single Gene Queries
 Gene Set Queries
Gene Set Queries
 Bulk Studies
Bulk Studies
 Single Cell Studies
Single Cell Studies
 Hypotheses
Hypotheses
 Resources
Resources
 Contribute
Contribute
 Downloads
Downloads About
About
 Help
Help