 Gene counts are sourced from ARCHS4, which provides uniform alignment of GEO samples.
You can learn more about ARCHS4 and its pipeline here.
Gene counts are sourced from ARCHS4, which provides uniform alignment of GEO samples.
You can learn more about ARCHS4 and its pipeline here.
Select conditions below to toggle them from the plot:
| GROUP | CONDITION | SAMPLES |
|---|---|---|
| Retinal neovascular proliferative membrane |
GSM2739339 GSM2739340 GSM2739341
|
|
|
GSM2739361 GSM2739362 GSM2739363
|
||
|
GSM2739342 GSM2739343 GSM2739344 GSM2739345 GSM2739346 GSM2739347 GSM2739348 GSM2739349 GSM2739350 GSM2739351 GSM2739352 GSM2739355 GSM2739356 GSM2739357 GSM2739358 GSM2739359 GSM2739360
|
||
|
GSM2739367 GSM2739368
|
||
| retina |
GSM2739364 GSM2739365 GSM2739366
|
|
|
GSM2739353 GSM2739354
|
Submission Date: Aug 10, 2017
Summary: Proliferative diabetic retinopathy (PDR) is the advanced stage of diabetic retinopathy (DR), coupling with irregular neovascularization, and is the leading cause of blindness in working-age people; but the molecular mechanism of vascular differentiation in PDR remains poorly characterized. In our study, we obtained the transcriptome profile of neovascular proliferative membrane specimens from patients with PDR via high-throughput sequencing and advanced bioinformatics. Marker genes of neovascularization were validated and distinct gene expression patterns were formed of PDR compared with normal retina. We also discovered gene sets that were co-expressed with vascular endothelial growth factor A (VEGFA), including transcription factors (TFs) that dysregulate VEGFA. In particular, ETS transcription factors family could negatively regulate VEGFA. We also detected a set of genes related to PDR was dramatically changed from pre-mRNA to mature mRNA. In summary, our study firstly presented the profile of neovascular proliferative membrane and identified new marker genes and key regulators of VEGFA, which could contribute the molecular therapy of PDR in the future.
GEO Accession ID: GSE102485
PMID: 31574534
Submission Date: Aug 10, 2017
Summary: Proliferative diabetic retinopathy (PDR) is the advanced stage of diabetic retinopathy (DR), coupling with irregular neovascularization, and is the leading cause of blindness in working-age people; but the molecular mechanism of vascular differentiation in PDR remains poorly characterized. In our study, we obtained the transcriptome profile of neovascular proliferative membrane specimens from patients with PDR via high-throughput sequencing and advanced bioinformatics. Marker genes of neovascularization were validated and distinct gene expression patterns were formed of PDR compared with normal retina. We also discovered gene sets that were co-expressed with vascular endothelial growth factor A (VEGFA), including transcription factors (TFs) that dysregulate VEGFA. In particular, ETS transcription factors family could negatively regulate VEGFA. We also detected a set of genes related to PDR was dramatically changed from pre-mRNA to mature mRNA. In summary, our study firstly presented the profile of neovascular proliferative membrane and identified new marker genes and key regulators of VEGFA, which could contribute the molecular therapy of PDR in the future.
GEO Accession ID: GSE102485
PMID: 31574534
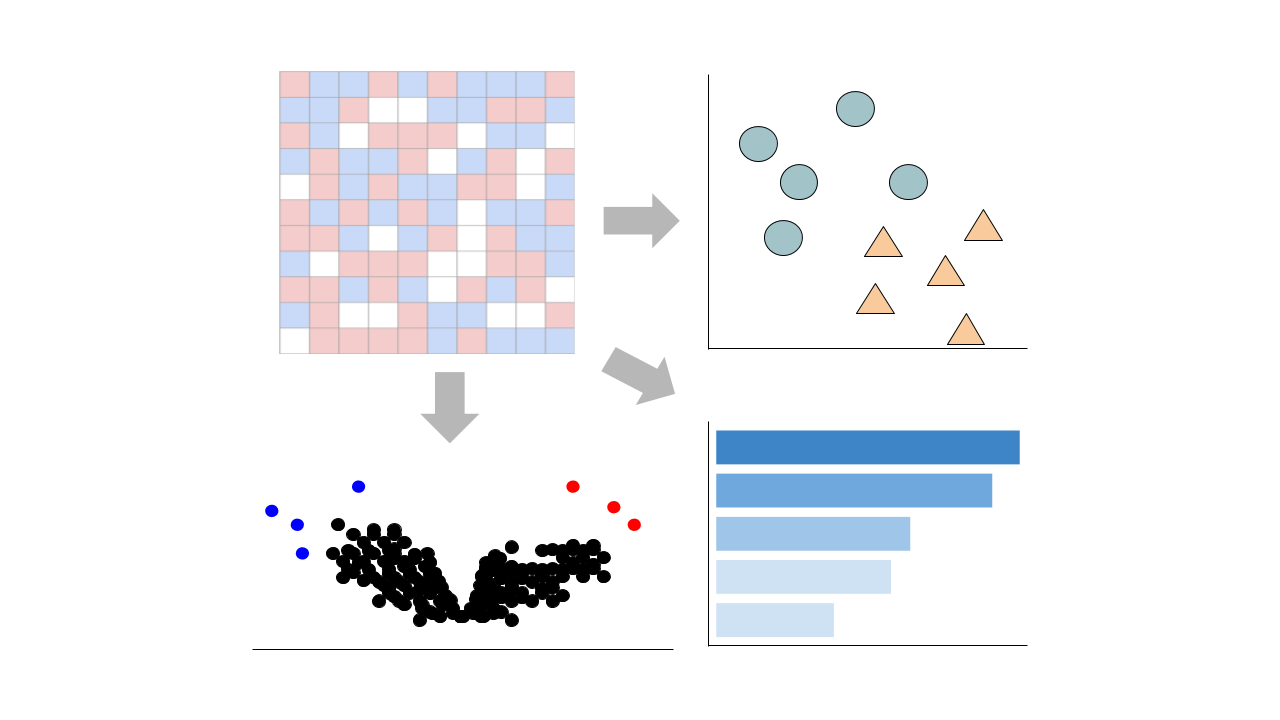
Visualize Samples
 Visualizations are precomputed using the Python package scanpy on the top 5000 most variable genes.
Visualizations are precomputed using the Python package scanpy on the top 5000 most variable genes.
Precomputed Differential Gene Expression
 Differential expression signatures are automatically computed using the limma R package.
More options for differential expression are available to compute below.
Differential expression signatures are automatically computed using the limma R package.
More options for differential expression are available to compute below.
Signatures:
Select conditions:
Control Condition
Perturbation Condition
Only conditions with at least 1 replicate are available to select
 Differential expression signatures can be computed using DESeq2 or characteristic direction.
Differential expression signatures can be computed using DESeq2 or characteristic direction.
This pipeline enables you to analyze and visualize your bulk RNA sequencing datasets with an array of downstream analysis and visualization tools. The pipeline includes: PCA analysis, Clustergrammer interactive heatmap, library size analysis, differential gene expression analysis, enrichment analysis, and L1000 small molecule search.
 Chatbot
Chatbot Single Gene Queries
Single Gene Queries
 Gene Set Queries
Gene Set Queries
 Bulk Studies
Bulk Studies
 Single Cell Studies
Single Cell Studies
 Hypotheses
Hypotheses
 Resources
Resources
 Contribute
Contribute
 Downloads
Downloads About
About
 Help
Help